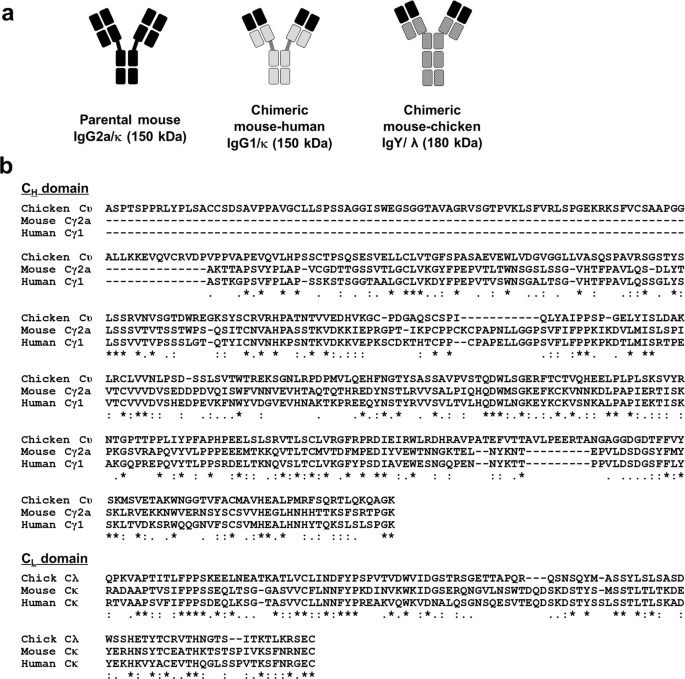
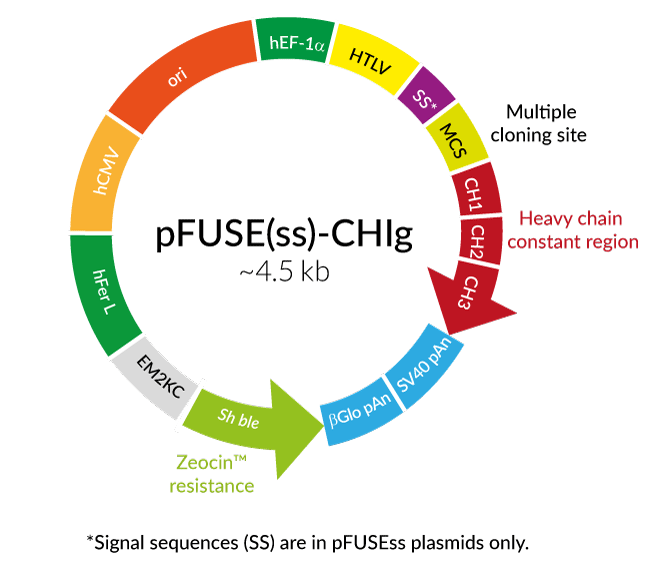
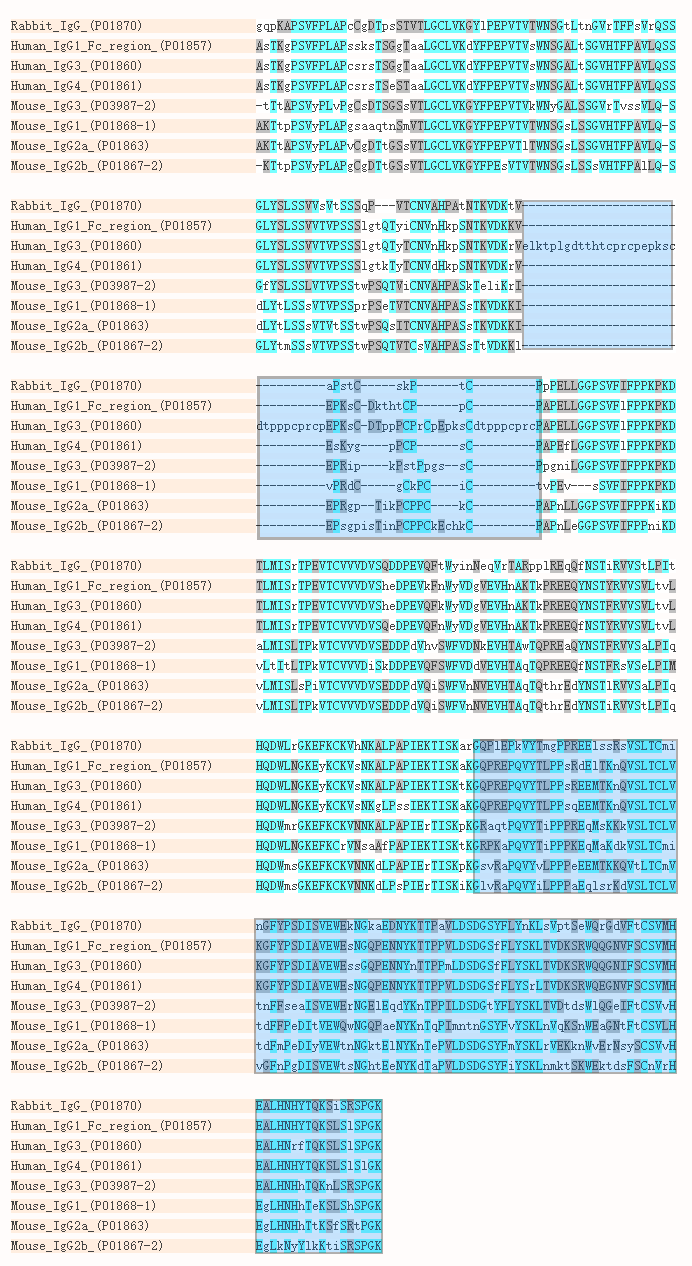
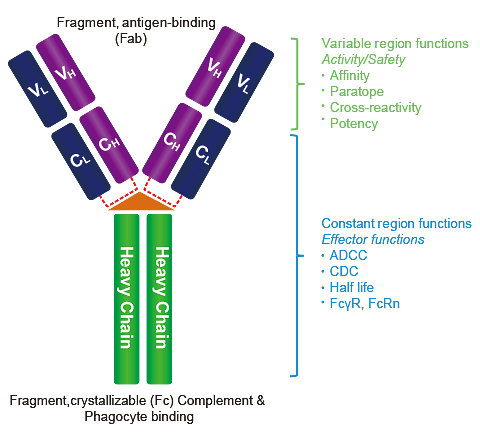
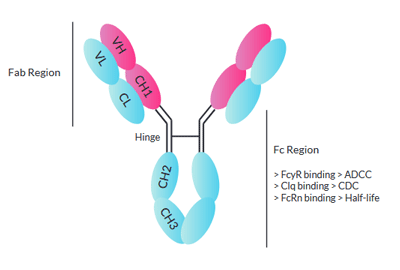
Amino acid sequence alignment of the Fc region of eqIgGs. The sequence... | Download Scientific Diagram

Structure of the streptococcal endopeptidase IdeS, a cysteine proteinase with strict specificity for IgG | PNAS

Relative Quantitation of Subclass-Specific Murine IgG Fc N-Glycoforms by Multiple Reaction Monitoring | ACS Omega

Figure 2 from Properties of mouse and human IgG receptors and their contribution to disease models. | Semantic Scholar
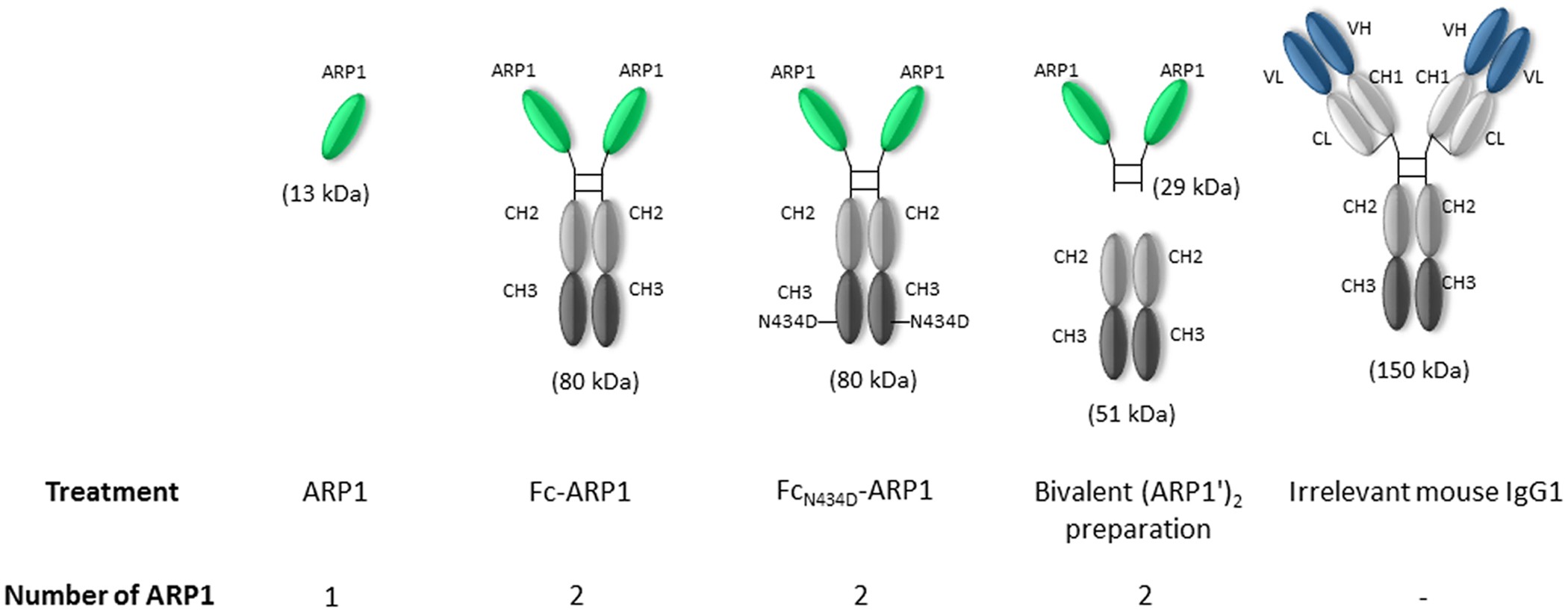
Fusion of the mouse IgG1 Fc domain to the VHH fragment (ARP1) enhances protection in a mouse model of rotavirus | Scientific Reports

Figure 1 from Functional, Biophysical, and Structural Characterization of Human IgG1 and IgG4 Fc Variants with Ablated Immune Functionality | Semantic Scholar

Engineering the hinge region of human IgG1 Fc-fused bispecific antibodies to improve fragmentation resistance | Scientific Reports

WO2018217988A1 - MODIFIED IgG1 Fc DOMAINS AND ANTI-CD40 DOMAIN ANTIBODY FUSIONS THEREWITH - Google Patents

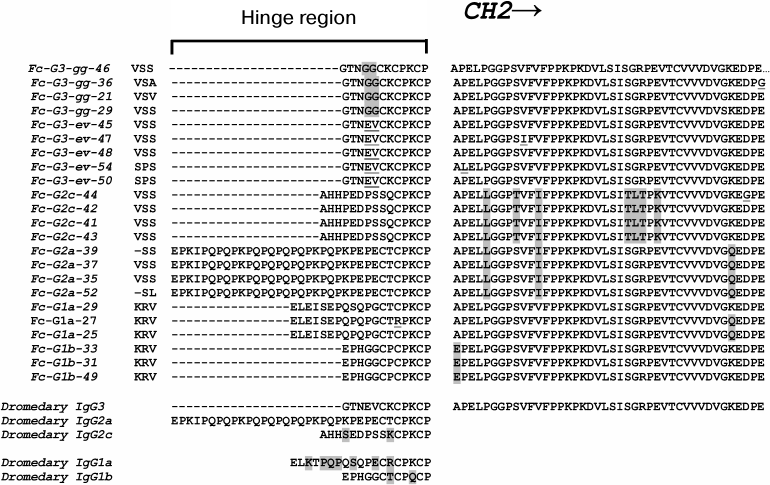
Analysis of heavy and light chain sequences of conventional camelid antibodies from Camelus dromedarius and Camelus bactrianus species - ScienceDirect

Structure-based Stability Engineering of the Mouse IgG1 Fab Fragment by Modifying Constant Domains - ScienceDirect